The FDA’s New Adverse Event Monitoring System (AEMS) Database
This Just In: Blog Post by AIM Chief Medical Officer Michelle Fiscus, MD
On March 11, 2026, the U.S. Food and Drug Administration (FDA) announced the launch of a new platform, the FDA Adverse Event Monitoring System (AEMS). This platform consolidates adverse event reports submitted to FDA for drugs, biologics, vaccines, cosmetics, and animal food. The announcement received very little attention considering the scope of the project. The FDA’s Chief Artificial Intelligence (AI) Officer described the project as “the biggest technical transformation in agency history.”
The FDA website states AEMS “will serve as a centralized platform for managing consumer complaints, regulatory misconduct reports, and whistleblower submissions across all FDA centers.” According to the FDA announcement, the following “legacy systems” have been “replaced” by AEMS:
- FDA Adverse Reporting System (FAERS) for drugs, biologics, cosmetics, and color additives
- Vaccine Adverse Event Reporting System (VAERS) for reports of adverse events after vaccination
- Adverse Event Reporting System (AERS) for animal drugs and animal foods
It is important to note, however, that the VAERS website, its reporting infrastructure, and reporting requirements have not changed with this migration to AEMS.
Additionally, the following legacy systems are slated to be replaced by AEMS in May:
- Manufacturer and User Facility Device Experience (MAUDE) for reports of medical devices
- Human Foods Complaint System (HFCS) for reports of human foods and dietary supplements
- Center for Tobacco Products Adverse Event Reporting System (CTPAE) for reports of electronic nicotine delivery systems (END) and other tobacco products
Along with the launch of the database, FDA has published an “AEMS Public Dashboard” that “is intended to expand access of AEMS data to the general public to search for information related to adverse events reported to the FDA by the industry, healthcare providers, and consumers.”
If you visit the AEMS dashboard site, you’ll first see a pop-up disclaimer that needs to be acknowledged that lists several considerations when interpreting AEMS data, including that “AEMS data alone cannot be used to establish event rates, evaluate changes in event rates over time, or compare event rates between products.” Once acknowledged, you can see the dashboard and its three tabs: Drugs and Biologics, Cosmetics, and Vaccines. When the Vaccines tab is selected, another pop-up disclaimer is shown that lists limitations of VAERS data and states, “reports to VAERS do not prove causation,” among other important details of the database. Once the disclaimer is acknowledged, you’re taken to the Vaccines dashboard:
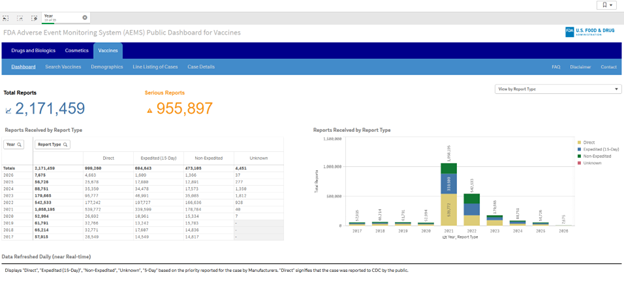
The main page lists total reports by year and denotes the total number of reports and “Serious Reports” at the top of the dashboard. Note, the “Direct” column represents cases reported directly to CDC by the public, whereas the other columns represent reports by manufacturers. From this dashboard, you can navigate to tabs that allow for searches by vaccines or adverse event terms. Per the website, dashboards are refreshed daily (“near real-time“). It is also possible to download a line listing of cases.
Again, it is important to note that the VAERS website, its reporting infrastructure, and reporting requirements have not changed. However, the move to display VAERS data through AEMS will have an impact on:
- How adverse events reports are viewed, since they are now publicly accessible in near real-time
- Who will use the data, since more people will likely interact with this system, leading to more claims of vaccine injury despite the disclaimers clearly stating AEMS data cannot be used for that purpose
- How users will interpret AEMS search results, which do not include significant narrative or outcome details, potentially causing users to draw incomplete or misleading conclusions about VAERS data
- The use of CDC Wonder, a database that has been historically used to query VAERS data, which still exists and now includes secondary, or follow-up, reports to an original adverse event report. This database contains more narrative and outcomes details than does the AEMS database but is more complicated to use
The bottom line: While the new AEMS dashboard provides greater public access to adverse events reporting and near real–time data, we can likely anticipate more references to VAERS data and claims of vaccine injury, regardless of the existence of evidence of causation.
